First, why do I care what the answer is? And why should you care? A little history may help you understand.
My son Jacob and I took on a project a little over three years ago, to convert my 1975 Triumph Spitfire roadster into a fully electric vehicle, which we call the E-Fire. The project flowed from my personal and professional interests in the transition to renewable energy sources and in reducing the environmental impact of global energy consumption. It was also spurred on by my purchase of our first Prius in 2008. I have driven nothing but Priuses since: I’m fascinated by the seamless way that Toyota managed to integrate the EV drivetrain with the Atkinson cycle gasoline engine. The vehicle not only has exceptional fuel economy, but toxic emissions are also greatly reduced.

(We won’t be needing THIS any more! 80 pounds of gasoline with a flip-top lid, separated from the passenger compartment by a 1/8″ thick sheet of pressboard covered with vinyl, always had me a little worried…)

(Jacob wiring the front battery pack: 22 LiFePO4 batteries. Another ten went where the gas tank used to be. The result is 100 km (60 miles) reliable range on a charge, without leaving the cells at a damagingly low depth of discharge)
The E-Fire project was a great opportunity for me to integrate these interests and to work together with my son at an age where he could be both a learner and a real helper too. That said, I would have considered the project a failure if I didn’t also produce a car which was useful for my commute, at a cost (ignoring our labour of course!) far less than that of the cheapest EV available to me at the time, a Gen 1 Nissan Leaf.

(If you’re going to put up with a 75 mile round-trip commute daily in the Toronto-Hamilton region’s notoriously disgusting traffic, you might as well do it in something fun!)
The result is spectacular: a car which is an absolute blast to drive, and which turns heads, but which has also reduced my commuting energy consumption by 80% and greenhouse gas emissions by 97%. That result is due to both the efficiency of the EV drivetrain and to Ontario’s unbelievable 40 g CO2/kWh electrical grid. After over 11,000 miles of driving, my face is still sore from the “EV grin”. The instant, quiet torque of an electric motor is quite intoxicating!
The project of course also fed into my interests in the feasibility and limits of both renewable generation and energy efficiency improvement measures by which we could meet the incredible challenge of transitioning away from fossil fuels- something which an honest evaluation of the science on the topic makes quite clear to be an absolute necessity. A lot of my LinkedIn commentary is now tied to issues related to efficiency, renewable generation and the threat of global warming, because frankly I see a tremendous amount of uninformed rubbish being spewed daily, in the form of unfair criticism or denial, unsubstantiated claims, crazy predictions or hype related to particular technologies. What often passes for “science journalism” in the Internet era just makes my blood boil: how can we expect non-technical people, which most of our voters and political leaders are, to make reasonable decisions on technical matters if we keep feeding them this garbage?
Case in point is the subject of this article: just exactly how much electricity does it take to refine a gallon of gasoline?
Here’s the quote from Elon Musk, in an interview related to the release of the movie “Revenge of the Electric Car”, which I found here:
Elon: “Exactly. Chris has a nice way of saying it which is, you have enough electricity to power all the cars in the country if you stop refining gasoline. You take an average of 5 kilowatt hours to refine gasoline, something like the Model S can go 20 miles on 5 kilowatt hours. You basically have the energy needed to power electric vehicles if you stop refining.”
Ding! My “hype detector” went off on that one, big time! This had to be an exaggeration. If it were true, it would make no economic sense at current retail gasoline prices, for one thing. But Elon Musk is a smart guy, so there’s no doubt a grain of truth in it, as there is in most myths. It was time for a little digging to find the source of the error.
I sat down and read the GM/Argonne National Laboratories well to tank and well to wheels studies to figure this out, as part of an effort to do accurate calculations for my own converted EV’s energy and GHG performance.
https://greet.es.anl.gov/publication-wft2tv3v
The GM/ANL study and the resulting GREET model are very complex, as you would expect given that oil refineries produce a lot of different fuels-gasoline, diesel and jet fuel, home heating oil and bunker fuel for ships, propane etc. But refineries produce a host of other products, and also internally recycle a lot of material and energy, often sharing energy and materials across the fence with numerous petrochemical plants which don’t even count as part of the refinery itself, and with the greater electrical grid.
The end result of a very careful and well-documented analysis via the GREET model which takes all of this complexity into account, is that from well to gas tank, gasoline is about 81 to 83% source energy efficient. The figures from the US average in 2001 were 98% for production/recovery, 84.5% for refining, 98.41% for distribution/transport to and from the refinery, and over 99.8% for storage. All the figures except the last one are from the GE/ANL well to tank study (2001) and subsequent well to wheels study, except the last one which is from Environment Canada. The figure compares almost exactly with the more recent EU JRC well to tank study (2014) which gives a comparable figure of about 82%. Put simply, this means that 118 J of energy in crude oil is used to produce 100 J of energy in the form of gasoline. 18 J is “lost” in the process.
As an engineer, such a comparison leaves me a bit cold. Even though heat and chemical energy and thermodynamic work and electrical energy all have the same units (Joules), that does not mean these forms of energy are all equivalent! The gods of thermodynamics take their tithe any time energy changes form.

OK, let’s look at Elon’s claim on its face: 5 kWh/US gallon is about 5/35.3 kWh or about 14% of the lower heating value (LHV) of a gallon of gasoline- but only if you were to convert the LHV (heat units) into electrical energy (kWh) at 100% efficiency. Ding! There’s the error! Of course aside from small home or jobsite generators, nobody burns gasoline to make electricity (regrettably there is still a lot of diesel burned though…). In general, simple fuel-burning power plants are on the order of 30% efficient based on the lower heating value (LHV) of the fuel. A modern gas-fired, combined cycle power plant can now reach about 60% efficiency, but that’s really not a fair comparison here.
Looking at the biggest energy sink in the gasoline production chain, which is refining at roughly 15% of the source energy, it’s important to know that most of the energy used in the refinery isn’t electricity. Fuel gas is used either directly in fired equipment or indirectly to produce steam, and it represents most of the source energy loss. A lot of that fuel gas in most refineries consists of byproduct streams from within the refinery itself, so it really does originate from the source, i.e. from crude oil itself.
Converting 15% of the chemical energy in a gallon of gasoline to electrical kWh at 100% efficiency is disingenuous because doing so in reality is impossible. On average, only about 15% of the energy used in a refinery is used in the form of electricity. Some refineries are electrical importers, and some are exporters from their cogen facilities, so only the average across a country or region is worth considering.
For a fair evaluation of Elon’s claim, the 15% of the 15% of the source (crude oil) energy used in the refinery in the form of electricity should be converted from source energy (fuel gas) at closer to 30% efficiency. A barrel of oil equivalent has a LHV of about 35.6 kWh/gal which is very close to the 35.3 kWh/gal LHV figure for a typical gasoline. So 15% x 15% x 35.6 kWh/gal is about 0.8 kWh/gallon of actual electrical energy use. Converted back to equivalent fuel gas used to make that electricity at 30% efficiency, you end up with an overall real well to tank efficiency for gasoline of about 80%, i.e. 2-3% worse than the figure which treats electric energy and fuel LHV as if they were equivalent. The GHG emissions figures in GREET work out to roughly the same result when you back-calculate.
My converted EV could drive about 3.2 miles at 250 Wh/mile on the electricity saved for every gallon of gasoline it didn’t use (that 250 Wh/mile figure is what my car averages, taking into account the efficiency of the charger and battery). Pre conversion, my Spitfire got about 29 miles to the US gallon if driven conservatively, so we’d make it roughly 1/10th as far. Elon’s Model S is a much heavier and larger car, so it consumes more like 400 Wh/mile and would only travel about 2 miles on that energy. Elon’s estimate is therefore out by an order of magnitude, and my “hype detector” was proven to be reasonably well calibrated.
We could do the more ridiculous comparison, taking the overall 20% of the source crude energy lost between well and tank, converting it all back to kWh at say 30% efficiency. That yields 20% x 30% x 35.6 kWh/gallon = 2.1 kWh, or enough electricity to take Elon’s model S a whopping 5.2 miles. Even on this ridiculous basis, Elon’s estimate is still out by a factor of about four.
Clearly, the amount of electricity used in refining and distributing gasoline is not insignificant, but nowhere nearly as high as Mr. Musk claimed. We’re going to have to find renewable sources of electricity if we want to convert all our gasoline cars to battery EVs- much less all the diesel cars, aircraft and ships dependent on crude oil. And more importantly, we have to remember that even if we decide tomorrow that fossil gasoline and diesel, jet and bunker fuel are no longer needed in any quantity (and I guarantee you that we won’t do that tomorrow, or in two decades either!), we would still be running crude oil refineries, adjusting the refinery processes to make a different suite of products. We’ll need to do that to produce the host of other materials and products that are every bit as necessary to modern life as electricity, but which are far harder to make from renewable sources.
I’ll leave you with the measured and calculated energetic and environmental performance of my E-Fire versus itself prior to conversion, and versus my most efficient vehicle- my Prius C hybrid. The results should be crystal clear- in a region like Ontario with a good grid, EVs are an environmental godsend.
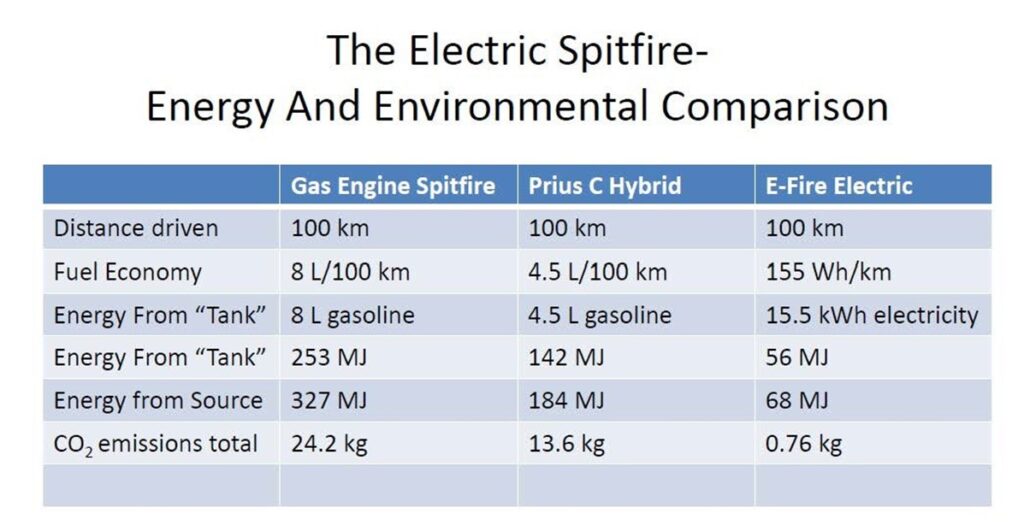
Battery EVs are a tremendous technology- one that will greatly help us toward a future which allows us to enjoy most if not all of the benefits we’ve derived from fossil energy, but without the GHG consequences. They will allow us to retain the freedom of individual transport, without discharging toxic emissions directly into the breathing zone of passersby. And the efficiency of the EV drivetrain and lithium-ion battery combination mean that we won’t have to build out as much expensive renewable infrastructure as we would if we were going to rely on other, less efficient renewable fuels such as biofuels or hydrogen- fuels which require more lossy steps of chemical or energy conversion. EVs don’t need hype or exaggerated claims to make them out to be more than they are: they can definitely stand on their own merits.